Pharma RCA Software for Root Cause Identification and Prevention
EviView’s platform offers a range of features to support operational teams, including Root Cause Analysis (RCA), which helps identify and address production challenges. By enabling teams to analyze issues and uncover actionable insights, EviView fosters proactive management, reduces inefficiencies, and enhances overall quality control within daily operations.
By providing actionable insights through advanced analytics, EviView empowers pharmaceutical manufacturers to minimize downtime, enhance quality control, and maintain seamless production. Shift from reactive problem-solving to proactive issue prevention with our intuitive RCA tools.


Effortless Root Cause Identification in Pharma Manufacturing
EviView simplifies the process of identifying the root causes of production issues by leveraging data-driven analytics and real-time monitoring. This ensures that your team can quickly pinpoint problems and implement effective solutions.
This includes:
- Data Consolidation: Integrate and centralize production data for a unified view of processes.
- Issue Detection: Use automated alerts and reports to identify deviations and trends early.
- Faster Resolution: Reduce the time it takes to investigate and resolve recurring issues.
EviView allows you to address problems as they arise while building long-term strategies for consistent operational excellence.












Beyond Basic Monitoring: EviView for Root Cause Analysis
EviView provides pharmaceutical manufacturers with advanced tools that go beyond basic troubleshooting. Its RCA capabilities are designed to foster a culture of continuous improvement and proactive risk management.
By providing clarity and direction, EviView’s root cause analysis software supports faster resolution and smarter prevention strategies.
Visual Analysis Tools
Use interactive diagrams, such as Fishbone and Pareto charts, to visualize root causes.
Actionable Insights
Generate detailed reports that guide your team in implementing corrective actions.
Collaborative Workflows
Share insights across departments to streamline problem-solving efforts.
Trend Identification
Monitor recurring patterns to address systemic issues proactively.
Centralize and Streamline RCA Data
EviView centralizes all RCA-related data into one secure platform, making it easier for teams to access, analyze, and act on critical information.
Centralized RCA data not only enhances operational transparency but also builds a foundation for sustained improvement.
Automated Data Collection
Capture real-time data from all production systems for seamless analysis.
Unified Storage
Maintain a single source of truth for all RCA documentation, improving consistency and accessibility.
Customizable Dashboards
Tailor data views and reports to suit the specific needs of your operations.
Secure and Compliant
Ensure sensitive production data is protected with robust security measures.
Optimize Production with RCA Insights
EviView’s RCA software equips pharmaceutical manufacturers with tools to optimize every aspect of their operations.
Proactive Risk Management
Identify potential risks early and implement mitigation strategies.
Reduced Downtime
Minimize equipment failures and process disruptions through targeted improvements.
Enhanced Quality Control
Ensure consistent product quality by addressing root causes of defects.
Streamlined Audits
Provide regulators and stakeholders with clear, data-backed reports.
Continuous Learning
Use past RCA insights to refine processes and prevent repeat issues.
Explore EviView Solutions for Root Cause Analysis in Pharma Manufacturing
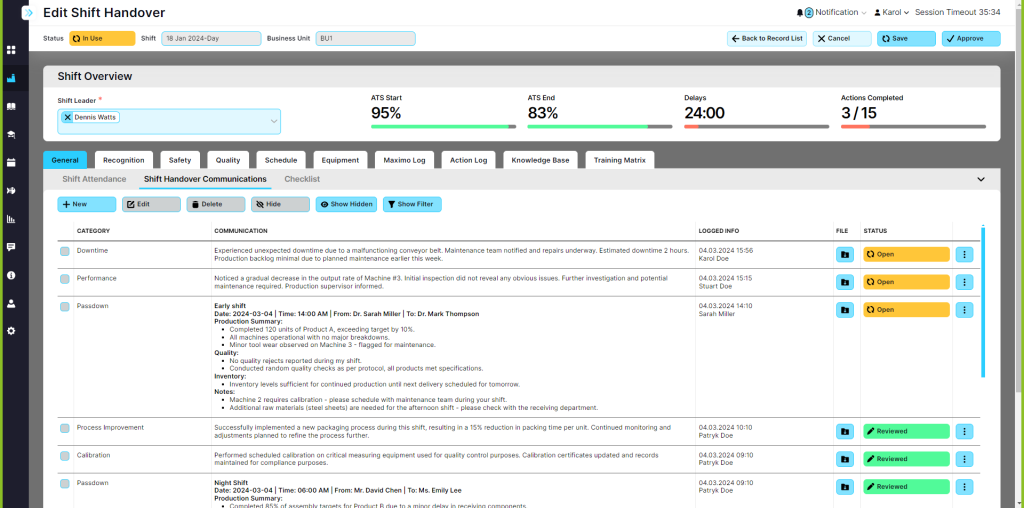
Streamline communication across shifts with intuitive handover tools, ensuring consistent safety and quality metrics.
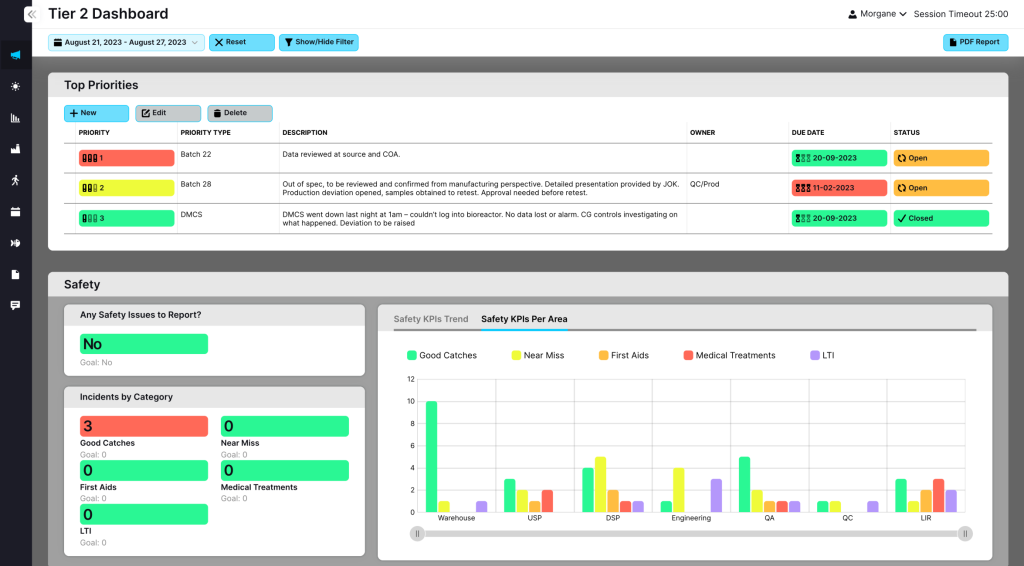
Gain real-time visibility into production KPIs, downtime analysis, and performance trends with powerful analytical tools.
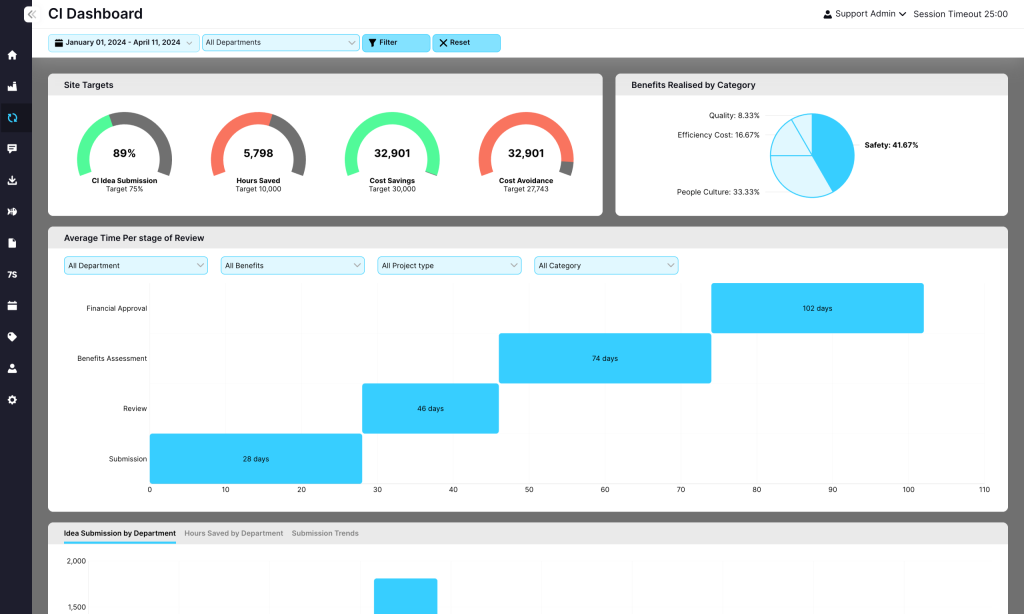
Empower teams to log and track improvement initiatives, supporting a culture of efficiency and innovation.
Take the First Step
A member of our team would love to introduce you to our platform and answer any questions you may have.