Why Shift Handovers Are Critically Important for Safe Operations

A shift handover process is straightforward in concept: communicate information so tasks can continue safely and effectively from one team to another. But when it comes to execution, this simple process can quickly get complicated. Shift handover is a critical operational event that happens regularly, which is why it shouldn’t be overlooked when you are looking at areas where you can improve efficiency. A poor shift handoff doesn’t just impact the smooth running of continuous processes; it can be a safety hazard too. What’s at stake: catastrophic consequences of poor communication Many large-scale industrial accidents can be attributed to miscommunication during shift handovers. One of the many factors that led to the 1988 Piper Alpha disaster, considered to be one of the world’s worst offshore disasters, was the failure to communicate proper information during the shift handover process. The public inquiry report by Lord Cullen found that the incoming shift hadn’t been informed that the pressure safety valve had been removed and replaced by a blind flange. The staff then took actions without this knowledge, which initiated the disaster and caused a loss of 167 lives and an insured loss of £1.7 billion. The 2014 DuPont La Porte incident in Texas also had a failure during the shift handover process, as per the U.S. Chemical Safety and Hazard Investigation Board report. The staff relied on verbal communication to convey troubleshooting operations to the incoming shift, but the details of what was communicated remained unclear as there were no written logs. This incident led to four workers losing their lives and 11,000 kg of the chemical methyl mercaptan being released from the pesticide manufacturing plant. These are just two examples of major accidents in which shift handoffs were found to be a factor. Incidents like these don’t always happen due to unusual events or malfunctioning machines. A Health and Safety Executive report by the UK government found that across all the incidents they examined, poor shift handovers during planned maintenance work always increased the risk of something going wrong. When maintenance took place over a change of shift, shift handover communication was not given high priority, so key information was not captured. Decisions made without key knowledge transfer led to major accidents that were, at times, fatal. Best practices for shift handover processes So how can we ensure that we have the right procedures for shift handovers in place to reduce the risk of accidents? There has been extensive research completed in order to determine the best shift handover practices, with similar recommendations across industries. Whether you look at the handover practices for offshore facilities or for running the Mars exploration rover, the most common overarching theme you’ll find is effective communication. Here are some points to keep in mind when creating a high-quality shift handover process: Establish clear procedures for handovers Shift handoffs should never be a hasty afterthought. There should be a clear procedure that fits in seamlessly with the rest of the workflow. The important messages need to be highlighted so they are not buried under irrelevant details. There should be sufficient time for handovers to take place, as they are a safety-critical process that requires vigilant follow-through to decrease the risk of accidents. The time set aside for the shift handover process should also be flexible, so it can go on longer if needed, such as when there’s a longer shift or when staff members return from extended leave periods. Identify and pay extra attention to higher-risk handovers There are always some shifts that need extra attention, and these shift handoffs should be identified beforehand. These are the shifts where there’s an increased risk of things going wrong, even at the slightest miscommunication. High-risk shift handovers include but are not limited to handovers between experienced and inexperienced staff, and during any deviations from regular operations, such as maintenance. Encourage two-way communication, preferably face-to-face Handovers are conventionally seen as one-way communication, which can lead to gaps in understanding. It’s not enough to leave a list of tasks and instructions for the next shift. Staff from both shifts should be equal participants in the shift handover process, with the new shift having the freedom to provide feedback and ask any questions for clarification. Face-to-face communication has also been found to be more effective, but in cases where it’s not an option, enabling two-way communication through video conferencing or over the phone is essential. Include written communication along with verbal instructions Both verbal and written communication is important to reduce the ambiguity of the information, and by providing it twice, it reduces the likelihood of erroneous information being communicated. Something captured in a logbook might not be mentioned verbally, or vice versa. Over-reliance on any one communication channel can lead to the intended message being misunderstood or not being communicated during the shift handover process. Keep in mind the information needs of the incoming staff There can be an assumption by the team that’s conducting the shift handoff that the incoming team is operating with the same mentality that they also think and operate in the same way that they do. This is why handovers should capture the intent of the information being communicated to aid comprehension. The incoming staff shouldn’t just know that an activity has to be completed; they also need to be aware of why it’s being done and what can go wrong if it doesn’t happen. Emphasize the importance of handover procedures and build the communication skills of the staff Since shift handovers are a regular process, it’s likely that complacency might set in and adherence to procedures may drop. The necessity and importance of following the established shift handover procedures should be clearly made known to the staff doing them. They should also be trained to improve their communication skills for a clearer transfer of information. The team should also be encouraged to help refine the shift handover process so it can be modified to suit their needs better. With effective shift handoffs being a high priority,
How To Achieve Continuous Improvement in Manufacturing
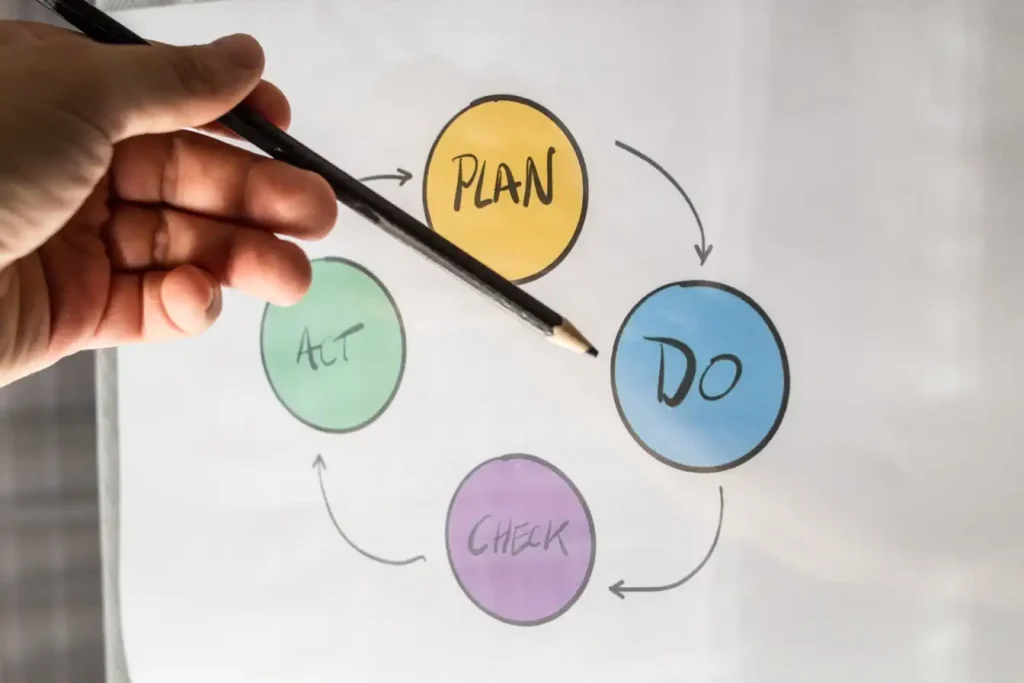
While efficiency has always been a top consideration for manufacturing companies, it is no longer optional. It is a must for business sustainability. The ripple effects of the pandemic have made it harder to achieve a profit. Consumer demand is fluctuating, supply chain issues still persist, and raw material prices are on the rise. While external factors cannot be controlled, manufacturers can optimize their internal processes. They can become more resilient and responsive by fine-tuning how they operate. This requires continuous improvement – implementing sustainable methods that make improvement initiatives not just a quarterly activity but rather an everyday part of your business. Understanding the principles of continuous improvement Continuous improvement models are behind many process management systems used by organizations, including lean production and Lean Six Sigma. This is also referred to as Kaizen, which in Japanese translates to good improvement. Businesses worldwide implement Kaizen as a pillar of their long-term competitive strategy. It can also be a highly beneficial manufacturing process improvement methodology. As per the Kaizen Institute, continuous improvement is made up of five fundamental principles. Here’s what manufacturers should know about them: Know your customer: Manufacturers should be aware of the demands of the customers for whom they are making the product, so they know exactly where they are creating value. Let it flow: Aim to create as little waste as possible so resources are used effectively during production. Go to GEMBA: GEMBA is a Japanese term that means “the actual place”. Executives should be aware of what’s happening at the place where manufacturing is actually done—the production floor. This is what’s behind the concept of a GEMBA walk, where executives visit the floor, see how operations are carried out, and talk to the workers. Empower people: Teams should be organized in a way that supports continuous improvement in a manufacturing organization. The goals of all teams should align and not clash with each other, so everyone can work together to achieve more. Employees should also be equipped with the tools and systems they need to reach these goals. Be transparent: Transparency here refers to data transparency. Through performance tracking, it is easy to see the tangible results of any changes being implemented, which proves their value and can also be a motivating factor. Adopting continuous improvement might sound intensive upfront, but it pays off over time. Today, with the assistance of digital solutions, implementing continuous improvement across the organization and scaling it as you grow is easier than ever. How to adopt continuous improvement initiatives Continuous improvement initiatives are implemented by organizations by establishing methods that can be effectively used by employees at all levels. Here is how manufacturers can adopt them. Define specific objectives Start by evaluating all the areas of your organization to identify your target areas for improvement.Some questions you can ask to identify these include: – Which areas are currently undergoing change? – What are the areas where bottlenecks or delays occur? – What are the areas that don’t meet quality or performance expectations? – Which areas have the most market or financial impact? After identifying the areas, define your objectives for improvement within the specific areas. For example, reducing lead time, improving quality, or reducing downtime. With the objectives in place, define the key performance indicators (KPIs) you will be using to measure progress towards the objective. Define the continuous improvement process While defining the continuous improvement process, workers from all levels should be involved so everyone’s perspectives are incorporated. At this stage, the responsibilities of the continuous improvement processes should also be placed on specific people who can actively maintain and manage them. Communicate your initiative clearly To enable the culture of continuous improvement, manufacturers first have to communicate it to all employees to encourage its adoption. Employees should be aware of what continuous improvement is in manufacturing and how it can help. They have to be trained in the methods and tools to carry out the continuous improvement processes. This can also empower employees to make their own suggestions for improvement. These training sessions should be frequent to reinforce the principles within the organization. Measure the outcomes of your initiatives Continuous improvement is all about results. After implementing changes, they have to be assessed after a sufficiently long interval to judge whether the improvement initiative has been successful or not. If not, it has to be adapted. If it has been successful, it still requires follow-up to ensure that the changes are sustained and permanently incorporated. Digital tools can make continuous improvement more convenient to adopt for manufacturers, as they can automate some steps such as tracking and reporting. They also make it possible to make decisions based on real-time data, including data from the manufacturing processes, so any urgent actions can be taken right away. EviView is custom-built for companies that want to make continuous improvement integral to the manufacturing process. With EviView, your organization can: Track various KPI metrics through trackers relevant to your objectives Integrate with existing systems that are already collecting data to present all the relevant information in an easily accessible place Generate reports that can help you analyze how the implemented initiatives have performed Once implemented, you can expect to see significant results over time. Through our tool, our award-winning team has already helped several manufacturers reduce delays by up to 20% and achieve productivity gains of 5–10%. We also hold training sessions so all employees can be comfortable using the tool and continuous improvement can be effectively achieved. Our tool has been especially useful for process industries, such as helping companies with their pharmaceutical batch manufacturing. Get in touch with us for a demo and discover how we can help your organization make improvements that amount to a number of extra days of manufacturing per year.
Bringing Transparency to Data-Driven Manufacturing

With the rise of digital transformation and, consequently, data-driven manufacturing, data visualization, ownership, and transparency are needed at all levels of the organization. Often, what exactly happens on the shop floors is unknown to the senior management, creating an information “black hole” that prevents them from making informed decisions based on manufacturing analytics. While there have been efforts to increase transparency and turn it into value for decades, it’s now becoming a necessity to compete in the market and increase profitability. In this blog post, we explore the importance of data transparency in data-driven manufacturing and its impact on operational performance. Why transparency in data-driven manufacturing matters and how it impacts operational performance One of the major challenges to information transparency in a manufacturing organization has historically been a lack of integration. While data was being collected manually or through software in different areas of the company, there was no place to see a holistic overview and support data-driven manufacturing. The ability to integrate real-time data into business decisions is now becoming increasingly accessible for all manufacturing firms as Industry 4.0 technologies such as IoT become more economical. Metrics from various sources can be combined into actionable dashboards. When it comes to trying these emerging technologies, however, what made some companies hesitant was the capital investment required for digital transformations. While complete digitization for a company might be expensive, many companies already have many processes digitized and would prefer not to shift to completely new systems just for integrating data sources. But the good news is that a new tech stack isn’t required to bring data transparency into your manufacturing organization. Several modern Industry 4.0 solutions, including EviView’s e-ConnX, can integrate into your existing tech stack and leverage data-driven manufacturing. Data transparency can now provide visible results by improving operational performance. Getting information from the factory floor, including the quality of production batches and downtime data, is especially valuable for process manufacturers, who also have to prioritize multiple regulatory compliances and optimize performance. Information transparency in data-driven manufacturing will also enable your organization to become more agile by being proactive rather than reactively managing operations. You can evaluate costs, analyze process productivity, and identify areas of opportunity specific to the plant. Along with this, manufacturing analytics will enable you to monitor any changes in real time, anticipate their effects, and allocate resources accordingly to minimize negative impacts. How manufacturers can achieve transparency in data-driven manufacturing Identify information gaps and align data collection with your KPIs Digital transformation strategies to add data transparency to data-driven manufacturing should align with your overall business strategy. With data transparency, you can expect to see aggregate metrics from various sources and contextualize them with your KPIs (key performance indicators). The selection of the KPIs should include all relevant stakeholders, especially the front-line employees and their managers, as their involvement is needed for any successful transformation. Pick your data collection and analytics solutions The next step after figuring out which metrics you want to track is choosing the right industrial IoT solutions to help you measure and analyze them. It may be hard to find a custom solution to do it all that is not cost-prohibitive. This is where tools that integrate within your existing tech stack and connect with other standard industry applications come in. Check whether you can automate data collection from machines and processes to the degree that your KPIs require. You should then be able to view all the metrics across the organization, from different business areas and plant sites, within a dashboard. Leverage analytics to create value Having all the data in one place should now enable you to make data-driven manufacturing decisions that improve production efficiency. Process improvement will be possible as results can be objectively measured and the effectiveness of new innovations tested. Cause and effect will be easier to evaluate, so you can better allocate your resources, optimize maintenance, and improve production quality. You can expect to see your organization’s performance improve over time as you continue to fine-tune your operations based on data. Stay ahead of the game with transparency in manufacturing analytics data Data transparency empowers your organization to understand operational performance better and discover opportunities for continuous improvement. Partnering with the right providers that share your vision for growth can help you accelerate your growth. EviView’s digital transformation solutions have been built with the aim of shifting knowledge upwards. Our e-ConnX app can give you greater visibility into process performance to track the production process from start to finish. Purpose-built for the bio, pharma, and chemical industries, our manufacturing analytics data solutions have the expertise of decades of R&D behind them. Get in touch with us today to learn how we can help you achieve the benefits of data transparency in data-driven manufacturing.
Operational Excellence in Pharma Manufacturing with Lean Six Sigma

While pharmaceuticals are a booming sector, in this increasingly competitive economy, companies have to constantly improve their manufacturing efficiency to be profitable and capture a larger share of the market. Digital transformation and the adoption of IoT solutions are necessary, but none of these can succeed without paying attention to the underlying processes. Many pharmaceutical manufacturers suffer from a lack of process information with no clear visibility into what’s really happening at all levels of the company, especially on the factory floor. Process management is what distinguishes the market leaders from those who lag behind. While quarterly targets might seem like an area of focus to quickly increase revenue, short-term goals do not necessarily set up the business for sustained long-term success. However, if the focus shifts to improving processes, you can also expect to see consistent success in achieving goals as your organization focuses on the factors that affect the eventual outcome. While there are many process management principles pharma manufacturers can follow, one of the most effective methodologies that has seen success across industries, including the manufacturing sector, is Lean Six Sigma. What is Lean Six Sigma (LSS)? Also referred to as LSS, Lean Six Sigma is a combination of two process management strategies: lean and six sigma. Lean manufacturing has its roots in the Japanese manufacturer Toyota’s production system. Their philosophy focused on reducing wasteful or redundant activities and improving workflow to draw more value from what you do. Eliminating waste and improving customer experience were the core of their thinking, and from that, lean manufacturing evolved as a way to increase speed. Six Sigma aims to improve process quality through statistical analysis that helps identify and eliminate defects, along with reducing variation in process outputs. The defects are actually process outputs that do not meet the customer specifications, a type of quality check. According to the six-sigma process, defects can be reduced by reducing the number of steps involved. This can be done through cause-and-effect analysis done by tracking the 6Ms. The 6Ms of Six Sigma are: Method: specific regulations or policies causing a slowdown Mother Nature: environmental factors affecting the production Manpower: training new people Measurement: accuracy of the equipment used to take readings Machine: maintenance issues or availability of equipment Materials: issues related to sourcing and transporting raw materials Together, Lean Six Sigma and manufacturing can be a powerful way to achieve operational excellence. Lean’s speed combined with Six Sigma’s speed overcomes the weaknesses of both methodologies, providing a complementing system that can be easily implemented in pharmaceutical manufacturing. Applying Lean Six Sigma to Pharmaceutical Manufacturing Lean Six Sigma initiatives are not new to the pharmaceutical industry. However, as they have lost their novelty and there is a lot of misunderstanding around the purpose of the methodology, it is still not widely implemented, and many companies have yet to experience how it can improve their pharma manufacturing process. Benefits of Lean Six Sigma in Pharma Manufacturing Here are how the two main benefits of Lean Six Sigma affect pharmaceutical manufacturing: Increased Speed Lean Six Sigma can be used to increase R&D productivity as well as speed up the production process. With a faster cycle time, pharmaceutical manufacturers can be the first to market and deliver products to their customers quickly. Improved Quality By standardizing processes through Lean Six Sigma, the variations in the drugs across batches can be reduced, and overall quality can improve. It can thus help reduce operational costs as you examine factors affecting formulation and quality closely, eliminating any wasteful activities that do not positively contribute to this. There are also recent applications of Lean Six Sigma that show that this methodology continues to be relevant and can yield great results. A 2021 study examined how a single pharmaceutical manufacturing company producing acetaminophen (paracetamol-containing pain relief) tablets used LSS to match the increased demand due to the pandemic. Operational excellence was achieved by using the toolset provided by this methodology to determine root causes and implement corrective actions. Problems were eliminated without negatively affecting manufacturing cost, production time, or product quality. The result was a reduction in downtime, improved product flow, the elimination of product waste, increased productivity, and an enhanced customer experience. This shows that Lean Six Sigma can help pharmaceutical companies achieve excellence, even in modern operations. Implement Lean Six Sigma with EviView As an award-winning Lean software provider, EviView has developed a one-stop solution for the process management needs of pharmaceutical manufacturing. Our tool helps your team seamlessly capture any delays categorized by the 6Ms (man, machine, material, method, measurement, and management) so you can adopt the Lean Six Sigma methodology. Book a call with us today to learn more about how you can achieve operational excellence.
Tracking and Improving Production Performance with EviView

Manufacturers have faced considerable production difficulties in recent years. As supply chains are disrupted, workforce dynamics change, and consumer demands evolve, manufacturers look for ways to optimize production efforts. Analyzing production data to identify patterns across the prevailing business landscape is one of the best ways to find optimization opportunities within a company. Manufacturers are leveraging data analytics to understand the forces influencing their operations and improve processes. Therefore, manufacturers have been increasingly investing in new digital solutions that help them gather more data from their business operations. With this, manufacturers can gain insight into how their machines function and adjust for optimum results. In this post, we explore some of the most common challenges plant leaders face and how you can leverage EviView business intelligence software to streamline data collection and draw more meaningful insights from your operations. Tracking the performance of plant operations A plant manager must know and understand the state of his plant’s performance to make better and more timely decisions. Unfortunately, most business leaders need more data they consider valuable. Despite this, they still need to track critical aspects of their operation and monitor performance trends to optimize decision-making as quickly as possible. Companies are also dealing with a shortage of skilled analytical professionals, making it exceedingly difficult for people outside their organization to understand and solve problems within their operations. This has led to a situation where many plant managers need to be more under- and over-managed. They need more information than they can access, but they also don’t want too much information because it can be overwhelming. They often feel like they’re fighting an uphill battle against their organizations, which makes sense considering that most companies lack the necessary tools to analyze operations data. Manufacturing challenges faced by plant leaders Manufacturing is a tough business, and plant leaders face several daily challenges. Here are some of the most common: Inefficient processesManufacturing companies are always working to improve their processes. They’re always looking for ways to improve efficiency and reduce costs, so manufacturers have a lot of data to analyze. The problem is that this data is often difficult to understand, which makes it hard for plant managers to know what’s causing problems or where they need to focus their attention. Poor data integrityData integrity is a big issue in manufacturing. The issue is that there are many data sources that may not always be accurate or up-to-date. For example, if you’re using an ERP system to keep track of sales orders and shipments but your shipping department has its own way of recording information about customer shipments, it’s easy for errors to creep into your data. Burdensome validationValidation is another big issue in manufacturing. You might have a lot of different applications that are all trying to get data from various sources into one place, but more is needed to ensure accuracy. You also need to ensure that the data you’re getting is accurate by validating it against other production monitoring systems or manually checking for errors. Unplanned deviationsIf you’re trying to automate your operations, it’s important to remember that there will always be unplanned deviations from the norm. The most obvious example is when a worker gets sick or injured, but other circumstances can cause problems. For example, if your equipment breaks down unexpectedly, it could throw off your schedule and create issues with production. Low in-process visibilityIn-process visibility refers to how well you can see what’s happening in each stage of your production process. If you have good visibility into the steps being performed, it can be easier to make changes when issues occur. This is especially true if one step relies on another to be completed before it happens, so if something goes wrong in the first stage, then there may not be anything that can be done about it until later (by which time it might already be too late). Delayed releaseDelayed releases are a common problem in manufacturing because they can cause other parts of the process to fall behind, leading to more delays and even worse quality problems. If you’re waiting on materials or other inputs from suppliers that need to meet your deadlines, it will throw off the entire schedule and create issues with production. Leveraging EviView for your plant operations EviView’s mission is to empower manufacturing monitoring with smarter, more effective manufacturing production tracking software for KPI tracking and reporting. We understand that the manufacturing industry is full of complex processes and production monitoring systems, making it difficult to get a clear picture of your plant’s performance. Behavioral change via continuous improvemente-ConnX makes it easy for operators to put forward CI Ideas as part of the shift handover—when they see their proposal in action, they’re more motivated. Continuous Improvement ideas can be submitted, reviewed, and plotted on the P-I-C-K chart. The more submissions employees make, the higher the chance of time and cost savings for a company. CI KPIs, such as average time to respond, review, and implement, are available in the module. Stakeholders can track the status of any actions originating from CI submission to implementation. Monitor issues efficiently with real-time data.EviView provides manufacturers with a view of their plants’ operations—a holistic, real-time look at key metrics that helps find bottlenecks, deviations, and downtime quickly. When a plant manager is notified of an issue or deviation, they can identify potential problems and prevent downtime by issuing early warnings to operators. It’s easy to monitor performance as it tracks all units minute by minute. Downtime and OEE scores can be seen for individual production lines as well as for the entire plant on a single screen in real-time. Data visualization for easier identification and quick resolutionEviView Analytics unlocks the power of your data to provide actionable insights that drive business-critical decisions. Using Fishbone, Ishikawa, Pareto, Waterfall charts, etc., clearly identifies which factors are the primary contributors to downtime events. When reporting downtime occurrences, operators can easily categorize and distinguish between issues caused