Biopharmaceutical Manufacturing, Protected

Safeguarding Bioprocess Consistency
Biopharmaceutical production depends on maintaining precise biological conditions over extended runs. Operators track culture behavior, scientists review sample results, and engineers monitor equipment performance as the batch progresses through each stage.
When these updates live in separate records or manual logs, teams struggle to see how the process is evolving. EviView brings operational notes, laboratory feedback, and process updates together in one system so every shift understands the true condition of the batch and the actions required to keep it on course.













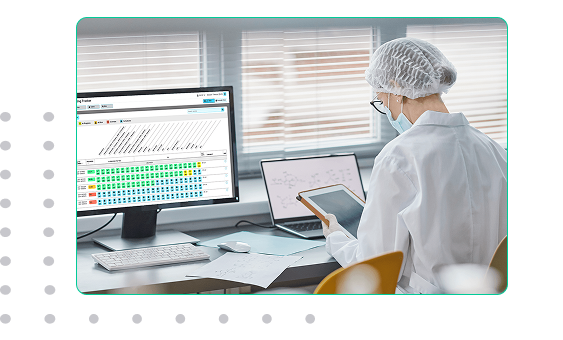
When Batch Context Is Lost, Investigations Begin
Biopharmaceutical batches generate a constant stream of observations, adjustments, and laboratory results. Over the course of a long run, dozens of small decisions shape the final outcome of the product.
When this information is scattered across notebooks, spreadsheets, and separate systems, teams struggle to reconstruct what actually happened during the process. EviView connects operational updates, process observations, and lab feedback to the batch record so teams can quickly understand events, respond to issues, and maintain control throughout production.
Reliable Execution for Biopharmaceutical Manufacturing
Keeping Every Batch on Track
Biopharmaceutical production depends on maintaining stable conditions across long fermentation and purification runs. EviView helps teams manage daily operations with clear shift communication, connected process updates, and shared visibility into the current state of the batch.
Batch Status in One Place
Operators record shift updates, process observations, and equipment conditions in one shared system.
Benefit: Teams always understand the current state of the batch before taking action.
Clear Handover Between Shifts
Important updates from the previous shift are captured and shared with the next team.
Benefit: Production continues without missing information or repeated checks.
Real Time Process Awareness
Production teams can view operational updates alongside MES, LIMS, and historian data.
Benefit: Teams detect process changes earlier and respond quickly.
Faster Deviation Investigation
Operational updates and events are recorded throughout the production run.
Benefit: Quality and production teams review the full history of the batch more quickly.
Key Capabilities for Biopharmaceutical Manufacturing
Batch-Centric Shift Logs
Link operational updates and actions directly to batch records.

Configurable Process Workflows
Adapt logs for cell culture, fermentation, purification, or fill operations.
System Integration
Connect MES, LIMS, and historian data with daily operations.
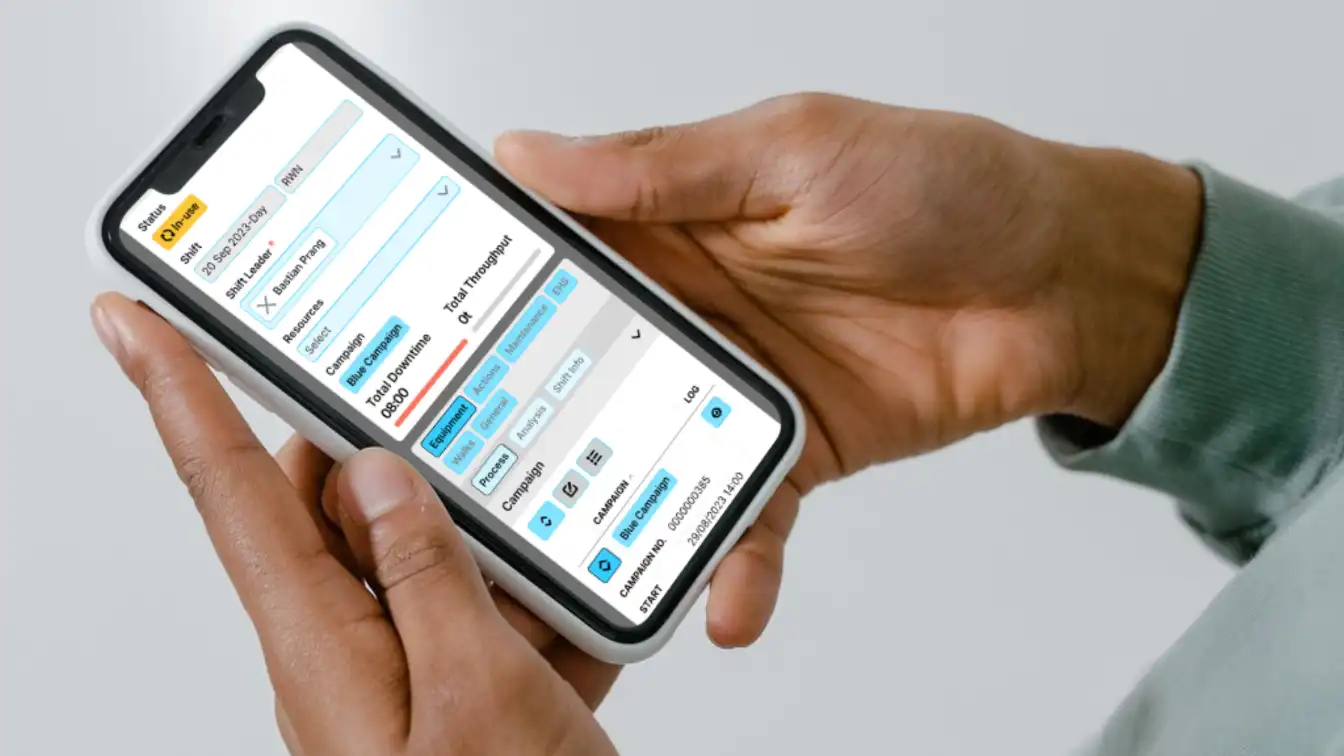
Digital Daily Management (DDMS)
Run Tier meetings with real production data and track actions.
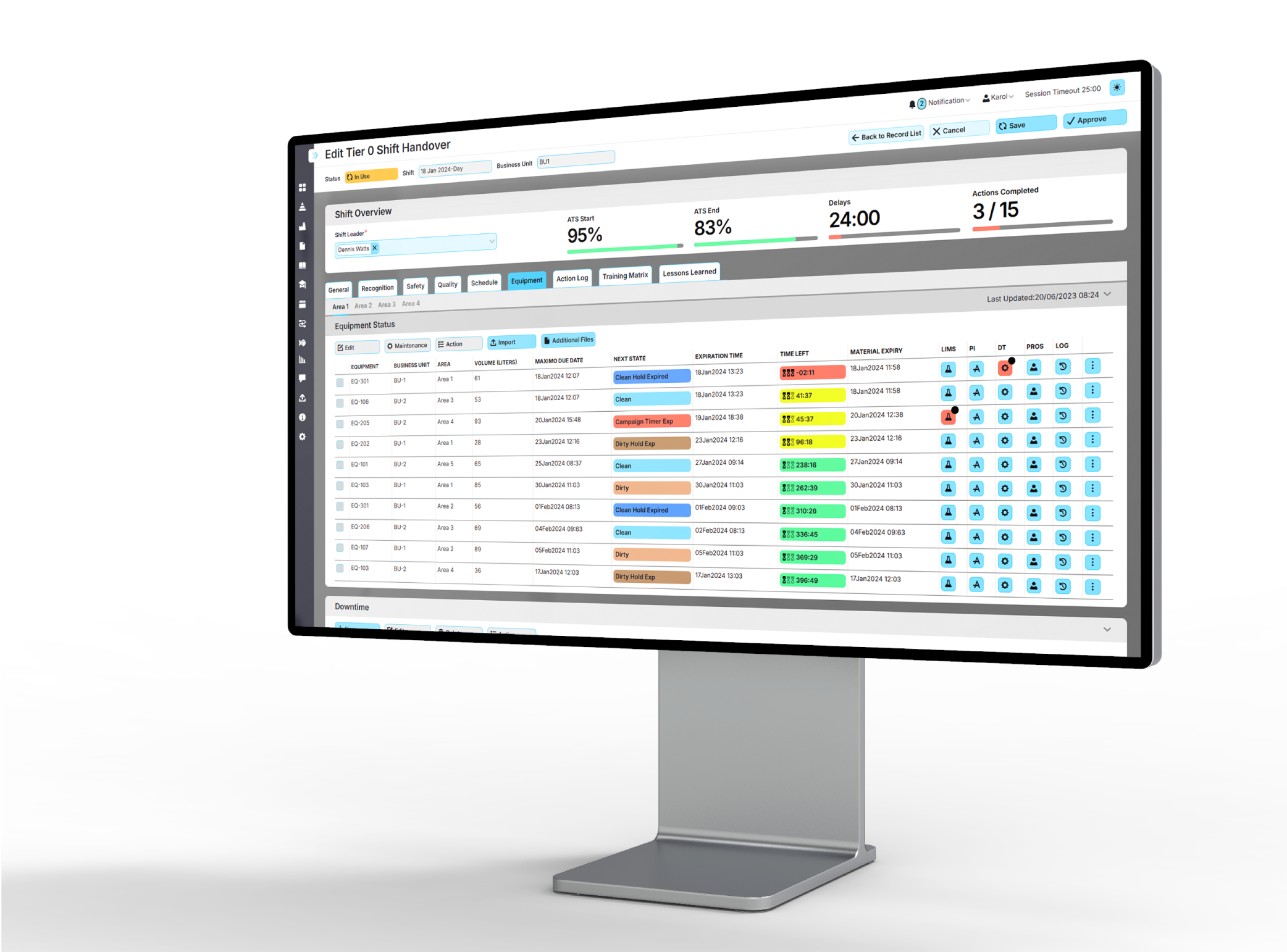
Secure Electronic Records
Maintain time-stamped records that support GMP documentation and review.

The Operational Impact for Biopharmaceutical Manufacturers
Biopharmaceutical manufacturers using EviView have achieved measurable improvements:
- Faster batch release cycles
- Up to 40% reduction in documentation errors and deviations
- Improved Right First Time (RFT) production performance
- Lower operational risk across long production runs
When information moves as quickly as the process itself, teams protect more than production output. They protect the patients who depend on these therapies.

Ensure Every Batch Reaches the Patient
Biopharmaceutical production is built on precision, coordination, and trust in every step of the process. EviView helps teams maintain control across long manufacturing runs so batches stay on track, deviations are addressed quickly, and critical therapies reach patients without delay.