Manufacturing Production Scheduling: A Path to Efficiency and Profitability

Manufacturing companies face a constant challenge: how to optimize production scheduling to meet customer demand while maintaining efficiency. This delicate balancing act can make or break a company’s success. In this blog post, we will explore the art of optimizing manufacturing production scheduling to enhance efficiency, reduce lead times, and ultimately increase profitability. Understanding Manufacturing Production Scheduling Manufacturing production scheduling is the art of planning and coordinating the production of goods within a manufacturing environment. It involves creating a detailed production plan that specifies the quantity and timing of each production task, along with the necessary resources for each task. The primary goal of production scheduling is to ensure that production runs efficiently, meeting customer demands while keeping costs in check. A production schedule typically comprises a task list, task timing, and resource requirements, encompassing materials, equipment, and personnel. Benefits of Optimizing Manufacturing Production Scheduling Optimizing manufacturing production scheduling can bring a plethora of benefits, revolutionizing a company’s operations and bolstering its competitive edge. Let’s take a closer look at these advantages: Improved Efficiency: Optimal production scheduling minimizes bottlenecks and idle time, allowing manufacturers to maximize productivity and output. This efficiency results in reduced production times and lower costs. Increased Profitability: Efficiency improvements translate into increased profitability. Reduced production times and costs, coupled with higher output, contribute to a healthier bottom line. Enhanced Customer Satisfaction: Meeting customer demand consistently and on time builds trust and satisfaction, fostering customer loyalty and brand reputation. Enhanced Quality Control: Streamlined scheduling reduces the risk of defects and ensures the consistency of products, enhancing overall product quality. Resource Utilization: Planning efficiently maximizes resource utilization, minimizes waste, and reduces costs. Greater Flexibility: Agile scheduling allows manufacturers to adapt to changing circumstances and market demands, reducing the risk of disruptions and delays. Conclusion In the dynamic world of manufacturing, mastering production scheduling is a critical component of achieving efficiency and profitability. By implementing effective scheduling strategies and embracing tools like production scheduling software, manufacturers can unlock these benefits and position themselves for success in an increasingly competitive market. As customer demands continue to evolve, optimizing production scheduling remains a vital driver of growth and profitability in the manufacturing sector.
The Cost of Bad Communication in Manufacturing Plants

In manufacturing, efficient communication is not just a desirable attribute; it’s an absolute necessity. The seamless flow of information within a manufacturing plant can significantly impact productivity, product quality, employee morale, and the overall bottom line. Conversely, the cost of bad communication can be staggering, both in financial terms and in terms of the long-term health of the organization. In this blog post, we will explore the various facets of this issue and understand why investing in effective communication is a crucial consideration for manufacturing plants. Production Delays One of the most immediate and obvious consequences of bad communication in a manufacturing plant is production delays. When critical information fails to reach the right people at the right time, it can lead to bottlenecks, downtime, and missed deadlines. Delays in production not only impact customer satisfaction but also increase operational costs as resources are underutilized and overtime pay may be required to catch up. Quality Control Quality control is paramount in manufacturing, and bad communication can undermine it significantly. Without clear communication channels, employees may not be aware of changes in production processes, quality standards, or product specifications. This can result in defective products, increased rework, higher scrap rates, and the potential for costly recalls or customer returns. Employee Morale and Retention Effective communication is not just about transmitting information; it’s also about fostering a positive work environment. When employees feel that their voices are not heard or that they are not well-informed about company policies and decisions, morale can suffer. It is detrimental to employee morale when employees don’t feel heard or are unaware of company policies and decisions. Consequently, it can lead to increased turnover rates, which come with their own set of recruitment and training costs. Safety Risks Safety should always be a top priority in manufacturing, and bad communication can pose significant safety risks. Failure to communicate essential safety protocols, hazards, or emergency procedures can result in accidents, injuries, and even fatalities. These incidents not only have devastating human costs but also legal and financial ramifications for the organization. Inventory Management Manufacturing plants often rely on efficient inventory management to minimize storage costs and maintain just-in-time production. However, when communication breakdowns occur between production and supply chain teams, inventory levels can become inaccurate. Overstocked or understocked items can lead to financial losses due to carrying costs or production halts when necessary materials are unavailable. Decision-Making In an era of data-driven decision making, timely access to accurate information is crucial. Bad communication can lead to confusion and misinformation, making it difficult for leaders to make informed choices. This can result in poor strategic decisions, missed opportunities, and financial losses in the long run. Reputation Damage Lastly, the cost of bad communication extends beyond the internal operations of a manufacturing plant. Negative publicity or customer complaints stemming from communication failures can damage the company’s reputation. Rebuilding trust and credibility can be a costly and time-consuming endeavor. Conclusion The cost of bad communication in manufacturing plants is multifaceted and can have far-reaching consequences. From production delays and quality control issues to safety risks and damage to the company’s reputation, the impacts are significant. To mitigate these costs, manufacturing plants should invest in robust communication systems, foster a culture of open and transparent communication, and provide training and tools to enhance communication skills at all levels of the organization. In doing so, they can not only reduce costs but also position themselves for long-term success in a highly competitive industry.
Leveraging Technology For Enhanced Performance Management

Staying competitive and efficient is vital for success. Leveraging technology has become a necessity, allowing organizations to make data-driven decisions, improve process visibility, and optimize operations. In this blog post, we explore how technology, particularly IT software and cutting-edge tools, is transforming performance management in manufacturing. We’ll look into the role of IT software, real-time data collection and analytics tools, automation, digitization, and the power of dashboards and visualizations in enhancing performance management. The Role of IT in Performance Management in Manufacturing IT software lies at the heart of modern performance management in manufacturing. With advanced software solutions, organizations can gather, analyze, and interpret data more effectively than ever before. Enterprise resource planning (ERP) systems and manufacturing execution systems (MES) are among the comprehensive tools available, offering capabilities for production scheduling, resource allocation, and quality control. By integrating various software solutions, manufacturers gain a holistic view of their operations, empowering them to make strategic decisions with greater precision. Utilizing Real-Time Data Collection and Analytics Tools Real-time data collection and analytics are game-changers in optimizing performance management in manufacturing. Thanks to sensor technology and IoT devices, manufacturers can capture data from machines, equipment, and production lines in real-time. This influx of real-time data allows for the swift detection of anomalies and deviations, enabling organizations to respond promptly and implement corrective actions. Advanced analytics tools process this data to identify trends, predict future performance, and uncover insights that were previously hidden in the vast amounts of data generated by modern manufacturing processes. Automation and Digitization of Performance Tracking and Reporting Automation and digitization play a crucial role in streamlining performance tracking and reporting, reducing manual effort and human errors. Tedious tasks such as data entry, report generation, and performance monitoring can be automated, freeing up valuable time for employees to focus on more strategic activities. Digitization of performance records ensures data accuracy, accessibility, and easy retrieval, facilitating seamless information sharing across departments and teams. Implementing Dashboards and Visualizations for Easy Data Interpretation Dashboards and data visualizations are indispensable tools for interpreting complex data sets quickly and efficiently. By presenting data visually through graphs, charts, and heatmaps, dashboards provide an easy-to-understand snapshot of key performance metrics. Manufacturing leaders can access real-time insights at a glance, facilitating faster decision-making and a rapid response to changing conditions. User-friendly dashboards allow stakeholders at all levels to drill down into data, enabling better collaboration and alignment across the organization. Conclusion Leveraging technology is no longer optional but essential for enhancing performance management in manufacturing. IT software, real-time data collection, analytics tools, automation, and digitization are revolutionizing how manufacturers monitor, analyze, and interpret performance data. These technologies empower organizations to optimize their operations, achieve higher levels of efficiency and productivity, and make data-driven decisions to remain competitive in an ever-evolving industry. By embracing technology as a key ally in performance management, manufacturing enterprises can unlock new opportunities for growth and success in the digital age. Embrace the future of manufacturing, where technology is your partner in achieving excellence.
Achieving Manufacturing Excellence Through Continuous Improvement and the PDCA Cycle

Continuous improvement is the cornerstone of successful manufacturing operations. In this blog post, we will delve into the world of continuous improvement in manufacturing, emphasizing the role of performance management as a catalyst for progress. We will also explore the importance of implementing a feedback loop for ongoing performance evaluation, the value of employee involvement in identifying and implementing improvements, and the power of the PDCA (Plan-Do-Check-Act) cycle as a tool for continuous enhancement. Continuous Improvement in Manufacturing Continuous improvement, often known as Kaizen in manufacturing, is the relentless pursuit of small but meaningful enhancements throughout the production process. It involves identifying inefficiencies, eliminating waste, and optimizing procedures to elevate productivity, quality, and customer satisfaction. Embracing continuous improvement empowers organizations to stay nimble, competitive, and adaptable in the face of market changes. Performance Management as a Driver for Progress Performance management is pivotal in steering continuous improvement within manufacturing. It comprises setting clear performance goals, regular monitoring of key performance indicators (KPIs), and analyzing performance data to pinpoint areas ripe for improvement. By aligning performance management with continuous improvement principles, organizations foster a culture that values data-driven decision-making, accountability, and the relentless pursuit of excellence. Implementing an Ongoing Feedback Loop A critical facet of performance management is establishing an open feedback loop for consistent performance evaluation. This entails regularly gathering and scrutinizing data to assess performance against predetermined targets and benchmarks. This feedback loop enables organizations to identify trends, spot improvement opportunities, and make timely adjustments to processes and operations. Leveraging technology and data analytics enhances the efficiency of the feedback loop, providing valuable insights to guide decision-making. Encouraging Employee Involvement Employees are the lifeblood of any successful manufacturing operation. Encouraging their active participation in identifying and implementing improvements is indispensable for sustained growth. Cultivating a culture that values employee ideas, fosters creativity, and empowers them to contribute to continuous improvement initiatives significantly impacts organizational performance. Regularly seeking input from frontline workers, who have an intimate understanding of the processes, can lead to innovative solutions and imbue employees with a sense of ownership. The Power of the PDCA Cycle The PDCA (Plan-Do-Check-Act) cycle, also known as the Deming Cycle, is a robust problem-solving methodology that supports continuous improvement efforts. Let’s delve into each stage of the PDCA cycle: PlanThe planning phase involves identifying improvement opportunities, setting specific goals, and creating detailed action plans. Stakeholder involvement ensures diverse perspectives and enhances the chances of success. DoDuring this stage, organizations implement proposed changes, typically on a small scale or in a controlled environment, to test their effectiveness. Effective communication and coordination among team members are crucial. CheckAfter implementing changes, organizations evaluate the results against established goals and KPIs. Data collection, analysis, and performance assessment are central to this stage. ActBased on the findings, organizations either standardize successful practices or refine action plans if outcomes fall short. The cycle then begins anew, encouraging a relentless pursuit of excellence. Benefits of the PDCA Cycle The PDCA cycle offers structured problem-solving, data-driven insights, adaptability, and employee engagement. It enables organizations to make informed decisions, standardize successful practices, and continuously refine their processes. When to Use the PDCA Cycle The PDCA cycle is adaptable across various industries and organizations. It excels at exploring effects in a controlled environment, minimizing waste through small-scale testing, and identifying the most effective improvements tailored to unique needs. When to use the PDCA cycle Conclusion The PDCA cycle is an ongoing process that becomes ingrained in an organization’s culture, guiding consistent improvement in products, services, or processes. Continuous improvement, driven by performance management and supported by the PDCA cycle, positions manufacturing operations for excellence, efficiency, and sustained growth in a dynamic business landscape. Embrace these principles, and your organization will thrive in the ever-evolving world of manufacturing.
Industry 5.0: Syncing Man, Machine and Methods

The evolution of technology and advancements in the industrial sector have paved the way for Industry 4.0 and Industry 5.0, also known as the Fourth and Fifth Industrial Revolutions. These new eras of manufacturing bring together physical and digital systems, resulting in smart factories that are fully automated and interconnected. Industry 5.0 is characterized by the integration of advanced technologies such as artificial intelligence, the Internet of Things, and robotics into manufacturing and production processes. For pharmaceutical and manufacturing companies, Industry 5.0 encapsulates the human element at its core, resulting in: Increased efficiency and productivityCompanies can use automation and AI to optimize production processes, reducing waste and errors. Improved product qualityAdvanced sensors and monitoring systems can provide real-time data on production processes, allowing for better quality control. Enhanced customer experienceCompanies can use advanced analytics and data processing to better understand customer needs and preferences, allowing for the development of more personalized products and services. Better supply chain managementCompanies can use IoT technologies to track products from raw materials to final delivery, improving transparency and reducing risks. SustainabilityIndustry 5.0 focuses on sustainability and the use of environmentally friendly technologies, reducing the negative impact of production on the environment. Industry 5.0 offers significant opportunities for pharmaceutical and manufacturing companies to improve their operations and stay ahead of the competition. However, with the increasing complexity of these new systems, it can be challenging to identify and fix errors that occur during the manufacturing process. This is where business intelligence software comes into play. In particular, EviView’s business intelligence software can help identify the root cause of errors and provide a smooth transition for stakeholders in Industry 4.0 and 5.0. Benefits of business intelligence solutions for manufacturing: Curtail lead and turnaround time EviView’s solution streamlines operations by reducing lead and turnaround times through efficient processes and data management. By automating manual tasks, the software saves time and reduces the chances of human error. Its centralized platform allows all departments to access relevant information in real-time, leading to faster decision-making and streamlined communication. These features result in shorter lead times, reduced turnaround times, and an overall increase in productivity and profitability. Place internal training controlsEviView places internal training controls in place by providing a comprehensive and organized platform for employee training and development. The software integrates all training materials, assessments, and feedback into one centralized system, allowing managers to track employee progress and monitor their understanding of the training. The software also provides custom reporting capabilities to help managers identify areas of strength and weakness in employee training and make informed decisions about future training plans. By implementing these internal training controls, EviView helps companies enhance the effectiveness of their training programs, improve employee performance, and drive business success. Streamline communicationsEviView’s software facilitates and syncs communication across departments by providing a centralized platform for collaboration and information sharing. It facilitates effective communication by allowing all departments to access and share relevant information, collaborate on projects, and track progress in real-time. This helps to eliminate miscommunication, reduce errors, and increase overall productivity and efficiency. By providing a comprehensive view of all projects and activities, our software helps to ensure that everyone is on the same page, resulting in better decision-making, improved teamwork, and greater success. Digital documentation With features like document scanning and OCR technology, data can be quickly captured and transformed into digital format, reducing the need for manual data entry. The software also includes powerful search and retrieval capabilities, making it simple to find the information you need when you need it. With EviView, organizations can improve accuracy, save time, and enhance their overall efficiency, making the transition from manual documentation to digital a seamless experience. IntegrationsThe software integrates with Industry 4.0 and 5.0 technologies, such as the Internet of Things (IoT) and artificial intelligence (AI), to provide a comprehensive view of the manufacturing process. This integration allows the software to collect data from a wide range of sources, including sensors, machines, and production systems, to provide a complete picture of the production process. One of the key benefits of using EviView’s business intelligence software is its ability to prevent errors from occurring. By identifying anomalies in the production process, the software can provide real-time alerts to stakeholders, allowing them to take corrective action before the issue becomes a bigger problem. For example, if the software identifies a machine that is producing subpar products, it can alert the maintenance team to fix the issue before it causes a production shutdown. This can save significant time and money, as well as prevent errors from being introduced into the final product. Furthermore, EviView’s business intelligence software provides a centralized platform for data analysis, allowing stakeholders to access real-time insights into the production process from one location. This can help eliminate the need for manual data analysis, saving time and reducing the risk of errors. EviView’s business intelligence tools include solutions ranging from digital shift handover to manufacturing analytics and continuous improvement that directly impact uniformity while reporting, accurate analysis, and efficient production process improvements.
How Lean Manufacturing Helps Pharmaceutical Facilities Reach Green Accreditations

As the world becomes increasingly conscious of the environmental impact of industrial activities, organizations across various sectors are striving to adopt sustainable practices. The pharmaceutical industry, in particular, plays a vital role in public health, but it is also under pressure to reduce its ecological footprint. One powerful approach proven to help pharmaceutical facilities achieve both operational efficiency and green accreditations is lean manufacturing. In this blog post, EviView CEO, Karol Dabrowski, explores the connection between lean manufacturing and green practices in the pharmaceutical industry, the challenges of implementing lean solutions, and the benefits of adopting this approach. The history behind lean manufacturing Although the idea of lean was first introduced in the 1450s by Henry Ford, Toyota is the most common name we associate with lean principles. Inspired by Ford’s production flow concept, Toyota created the ‘Toyota Production System’ in lean principles after World War II. The novelty about Toyota’s approach is its focus on the whole process and not just the machines themselves. A set of principles and practices focused on reducing waste in processes, improving efficiency, and maximising value for customers, lean manufacturing was designed to enhance overall productivity. The emergence of generic competition Ever since the introduction of new regulations in the pharmaceutical sector, which eliminated the lengthy testing requirements for producing generic drugs, competition rates have soared.While in 1983, only 35 percent of the top-selling drugs with expired patents had a generic competitor, this rate has risen and is closer to 100 percent nowadays. Generic versions emerge virtually quickly when a brand-name drug’s patent expires. Consequently, pharmaceutical companies find themselves in intense competition with generics and biosimilars and industry watchers forecast that from 2023-2030, the industry’s top drug companies will lose more than $200 billion. The biopharma industry is expected to face a number of high-profile patent cliffs between now and 2030, which are likely to reshape the market. As several prominent brands in the US and Europe prepare to lose market exclusivity and encounter competition from generics or biosimilars,the need for finding ways to reduce costs and invest in R&D becomes even more latent.. Consequently, the adoption of lean practices has become crucial for manufacturing facilities, enabling companies to identify waste and address manufacturing process issues that could affect profitability. Furthermore, as lean manufacturing tools and processes continue to evolve, notable trends are emerging, including a growing emphasis on sustainability, digital transformation, and the advent of IoT 5.0. The push for sustainable manufacturing and green accreditations Considered one of the biggest trends in lean manufacturing, sustainability has been a recurrent agenda item in the sector. Deloitte’s ‘Embedding Environmental Sustainability into Pharma’s DNA’ report outlines that the pharmaceutical industry’s participation in environmental challenges is multidimensional, having ramifications for the whole product value chain. However, most companies likely underestimate the size of the challenge, given the inherent complexities of the industry. Beyond simply promoting sustainable practices, pharmaceutical companies now face the need to take accountability and undergo a fundamental transformation through evidence-based improvement initiatives. The application of new processes, techniques, and technologies to reduce the environmental impact of industrial operations while maintaining or enhancing economic performance is what sustainable manufacturing entails. Green accreditations like LEED certification and ISO 14001 environmental management systems serve to recognize and reward organizations committed to sustainable practices. These certifications frequently require businesses to achieve specified standards for energy use, waste management, and resource efficiency, goals that align well with the principles of lean manufacturing. The connection between lean manufacturing and green practices Lean manufacturing and green practices share a fundamental objective: to eliminate waste and optimize resource utilization. By implementing lean principles in pharmaceutical facilities, organizations can address various environmental challenges associated with their operations. Here are some key connections between lean manufacturing and green practices: Waste Reduction: Lean manufacturing aims to eliminate all forms of waste, including excess inventory, overproduction, defects, transportation waste, and more. By reducing waste, pharmaceutical facilities can minimize the consumption of resources, energy, and raw materials, resulting in a lower environmental impact. Energy and Resource Efficiency: Lean manufacturing principles promote the efficient use of energy and resources by streamlining processes and eliminating non-value-added activities. This approach encourages facilities to adopt energy-saving technologies, optimize equipment usage, and implement sustainable practices, thereby reducing greenhouse gas emissions and conserving natural resources. Process Optimization: Lean manufacturing emphasizes continuous improvement and process optimization. By eliminating bottlenecks, unnecessary steps, and redundancies, pharmaceutical facilities can streamline their operations, reduce energy consumption, and minimize waste generation. The challenges of implementing lean solutions in pharmaceutical facilities While implementing lean solutions can bring significant benefits, pharmaceutical facilities may face certain challenges in the process. These challenges include: Compliance with regulatory requirements: The pharmaceutical industry is subject to stringent regulations and quality standards. Implementing lean solutions must ensure compliance with these requirements while simultaneously driving operational improvements and sustainability initiatives. Change management: Introducing lean manufacturing practices often requires significant changes to processes, workflows, and employee roles. Resistance to change and the need for training and cultural transformation can pose challenges during the implementation phase. Technological investment: Implementing lean solutions may involve investing in new technologies, equipment, and systems. Pharmaceutical facilities need to carefully evaluate these investments and ensure they align with sustainability goals and long-term benefits. Tackling the challenges Although many pharmaceutical companies recognize the importance of implementing lean manufacturing practices to achieve green accreditations, their attempts to adopt lean principles have often fallen short due to a lack of structure and inadequate support. Despite investing in training, without a solid framework in place, lean processes become unsustainable and can descend into chaos. Sustaining lean processes and fostering a culture of continuous improvement require the right tools. While the concepts of lean are understood, it is often a struggle to implement them effectively when everyone is already overwhelmed with their workload. This is where digital transformation comes into play, offering solutions that empower pharmaceutical facilities to manage and control manufacturing processes more accurately. By
How To Achieve Continuous Improvement in Manufacturing
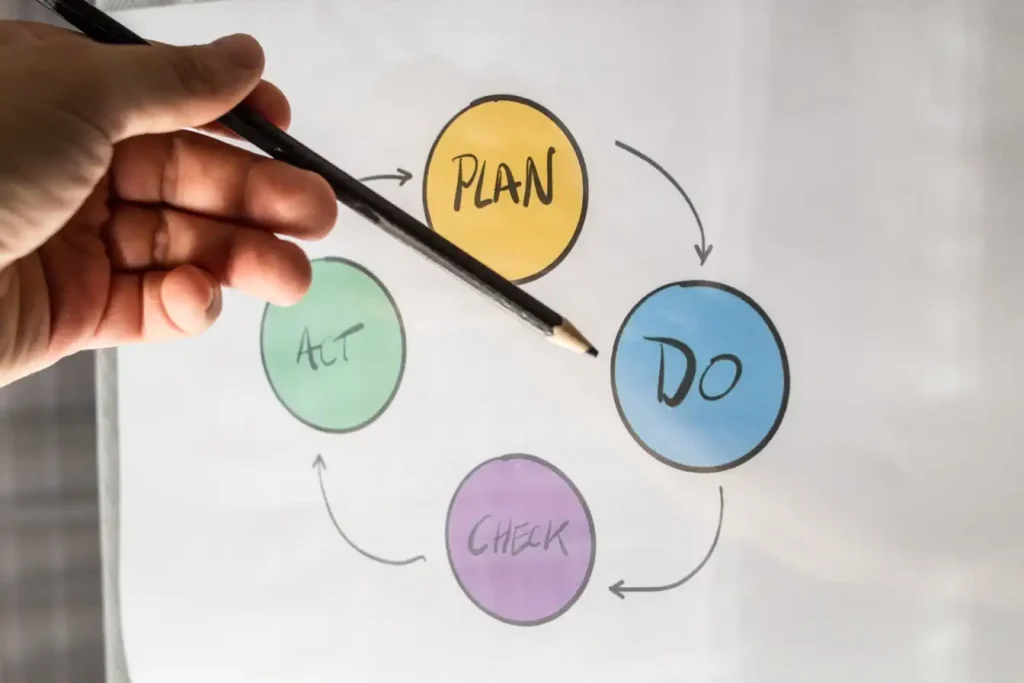
While efficiency has always been a top consideration for manufacturing companies, it is no longer optional. It is a must for business sustainability. The ripple effects of the pandemic have made it harder to achieve a profit. Consumer demand is fluctuating, supply chain issues still persist, and raw material prices are on the rise. While external factors cannot be controlled, manufacturers can optimize their internal processes. They can become more resilient and responsive by fine-tuning how they operate. This requires continuous improvement – implementing sustainable methods that make improvement initiatives not just a quarterly activity but rather an everyday part of your business. Understanding the principles of continuous improvement Continuous improvement models are behind many process management systems used by organizations, including lean production and Lean Six Sigma. This is also referred to as Kaizen, which in Japanese translates to good improvement. Businesses worldwide implement Kaizen as a pillar of their long-term competitive strategy. It can also be a highly beneficial manufacturing process improvement methodology. As per the Kaizen Institute, continuous improvement is made up of five fundamental principles. Here’s what manufacturers should know about them: Know your customer: Manufacturers should be aware of the demands of the customers for whom they are making the product, so they know exactly where they are creating value. Let it flow: Aim to create as little waste as possible so resources are used effectively during production. Go to GEMBA: GEMBA is a Japanese term that means “the actual place”. Executives should be aware of what’s happening at the place where manufacturing is actually done—the production floor. This is what’s behind the concept of a GEMBA walk, where executives visit the floor, see how operations are carried out, and talk to the workers. Empower people: Teams should be organized in a way that supports continuous improvement in a manufacturing organization. The goals of all teams should align and not clash with each other, so everyone can work together to achieve more. Employees should also be equipped with the tools and systems they need to reach these goals. Be transparent: Transparency here refers to data transparency. Through performance tracking, it is easy to see the tangible results of any changes being implemented, which proves their value and can also be a motivating factor. Adopting continuous improvement might sound intensive upfront, but it pays off over time. Today, with the assistance of digital solutions, implementing continuous improvement across the organization and scaling it as you grow is easier than ever. How to adopt continuous improvement initiatives Continuous improvement initiatives are implemented by organizations by establishing methods that can be effectively used by employees at all levels. Here is how manufacturers can adopt them. Define specific objectives Start by evaluating all the areas of your organization to identify your target areas for improvement.Some questions you can ask to identify these include: – Which areas are currently undergoing change? – What are the areas where bottlenecks or delays occur? – What are the areas that don’t meet quality or performance expectations? – Which areas have the most market or financial impact? After identifying the areas, define your objectives for improvement within the specific areas. For example, reducing lead time, improving quality, or reducing downtime. With the objectives in place, define the key performance indicators (KPIs) you will be using to measure progress towards the objective. Define the continuous improvement process While defining the continuous improvement process, workers from all levels should be involved so everyone’s perspectives are incorporated. At this stage, the responsibilities of the continuous improvement processes should also be placed on specific people who can actively maintain and manage them. Communicate your initiative clearly To enable the culture of continuous improvement, manufacturers first have to communicate it to all employees to encourage its adoption. Employees should be aware of what continuous improvement is in manufacturing and how it can help. They have to be trained in the methods and tools to carry out the continuous improvement processes. This can also empower employees to make their own suggestions for improvement. These training sessions should be frequent to reinforce the principles within the organization. Measure the outcomes of your initiatives Continuous improvement is all about results. After implementing changes, they have to be assessed after a sufficiently long interval to judge whether the improvement initiative has been successful or not. If not, it has to be adapted. If it has been successful, it still requires follow-up to ensure that the changes are sustained and permanently incorporated. Digital tools can make continuous improvement more convenient to adopt for manufacturers, as they can automate some steps such as tracking and reporting. They also make it possible to make decisions based on real-time data, including data from the manufacturing processes, so any urgent actions can be taken right away. EviView is custom-built for companies that want to make continuous improvement integral to the manufacturing process. With EviView, your organization can: Track various KPI metrics through trackers relevant to your objectives Integrate with existing systems that are already collecting data to present all the relevant information in an easily accessible place Generate reports that can help you analyze how the implemented initiatives have performed Once implemented, you can expect to see significant results over time. Through our tool, our award-winning team has already helped several manufacturers reduce delays by up to 20% and achieve productivity gains of 5–10%. We also hold training sessions so all employees can be comfortable using the tool and continuous improvement can be effectively achieved. Our tool has been especially useful for process industries, such as helping companies with their pharmaceutical batch manufacturing. Get in touch with us for a demo and discover how we can help your organization make improvements that amount to a number of extra days of manufacturing per year.
Bringing Transparency to Data-Driven Manufacturing

With the rise of digital transformation and, consequently, data-driven manufacturing, data visualization, ownership, and transparency are needed at all levels of the organization. Often, what exactly happens on the shop floors is unknown to the senior management, creating an information “black hole” that prevents them from making informed decisions based on manufacturing analytics. While there have been efforts to increase transparency and turn it into value for decades, it’s now becoming a necessity to compete in the market and increase profitability. In this blog post, we explore the importance of data transparency in data-driven manufacturing and its impact on operational performance. Why transparency in data-driven manufacturing matters and how it impacts operational performance One of the major challenges to information transparency in a manufacturing organization has historically been a lack of integration. While data was being collected manually or through software in different areas of the company, there was no place to see a holistic overview and support data-driven manufacturing. The ability to integrate real-time data into business decisions is now becoming increasingly accessible for all manufacturing firms as Industry 4.0 technologies such as IoT become more economical. Metrics from various sources can be combined into actionable dashboards. When it comes to trying these emerging technologies, however, what made some companies hesitant was the capital investment required for digital transformations. While complete digitization for a company might be expensive, many companies already have many processes digitized and would prefer not to shift to completely new systems just for integrating data sources. But the good news is that a new tech stack isn’t required to bring data transparency into your manufacturing organization. Several modern Industry 4.0 solutions, including EviView’s e-ConnX, can integrate into your existing tech stack and leverage data-driven manufacturing. Data transparency can now provide visible results by improving operational performance. Getting information from the factory floor, including the quality of production batches and downtime data, is especially valuable for process manufacturers, who also have to prioritize multiple regulatory compliances and optimize performance. Information transparency in data-driven manufacturing will also enable your organization to become more agile by being proactive rather than reactively managing operations. You can evaluate costs, analyze process productivity, and identify areas of opportunity specific to the plant. Along with this, manufacturing analytics will enable you to monitor any changes in real time, anticipate their effects, and allocate resources accordingly to minimize negative impacts. How manufacturers can achieve transparency in data-driven manufacturing Identify information gaps and align data collection with your KPIs Digital transformation strategies to add data transparency to data-driven manufacturing should align with your overall business strategy. With data transparency, you can expect to see aggregate metrics from various sources and contextualize them with your KPIs (key performance indicators). The selection of the KPIs should include all relevant stakeholders, especially the front-line employees and their managers, as their involvement is needed for any successful transformation. Pick your data collection and analytics solutions The next step after figuring out which metrics you want to track is choosing the right industrial IoT solutions to help you measure and analyze them. It may be hard to find a custom solution to do it all that is not cost-prohibitive. This is where tools that integrate within your existing tech stack and connect with other standard industry applications come in. Check whether you can automate data collection from machines and processes to the degree that your KPIs require. You should then be able to view all the metrics across the organization, from different business areas and plant sites, within a dashboard. Leverage analytics to create value Having all the data in one place should now enable you to make data-driven manufacturing decisions that improve production efficiency. Process improvement will be possible as results can be objectively measured and the effectiveness of new innovations tested. Cause and effect will be easier to evaluate, so you can better allocate your resources, optimize maintenance, and improve production quality. You can expect to see your organization’s performance improve over time as you continue to fine-tune your operations based on data. Stay ahead of the game with transparency in manufacturing analytics data Data transparency empowers your organization to understand operational performance better and discover opportunities for continuous improvement. Partnering with the right providers that share your vision for growth can help you accelerate your growth. EviView’s digital transformation solutions have been built with the aim of shifting knowledge upwards. Our e-ConnX app can give you greater visibility into process performance to track the production process from start to finish. Purpose-built for the bio, pharma, and chemical industries, our manufacturing analytics data solutions have the expertise of decades of R&D behind them. Get in touch with us today to learn how we can help you achieve the benefits of data transparency in data-driven manufacturing.
